About InLighta Biosciences
Leading the Future of
Precision Diagnostics

Meet Our Leadership Team
At InLighta Biosciences, we’re driven by a team of visionary leaders and talented professionals dedicated to revolutionizing medical diagnostics.

Dr. Jenny Yang
President and Founder
A leading figure in the field of molecular imaging, Dr. Jenny Yang founded InLighta Biosciences with a vision to transform the landscape of precision diagnostics.
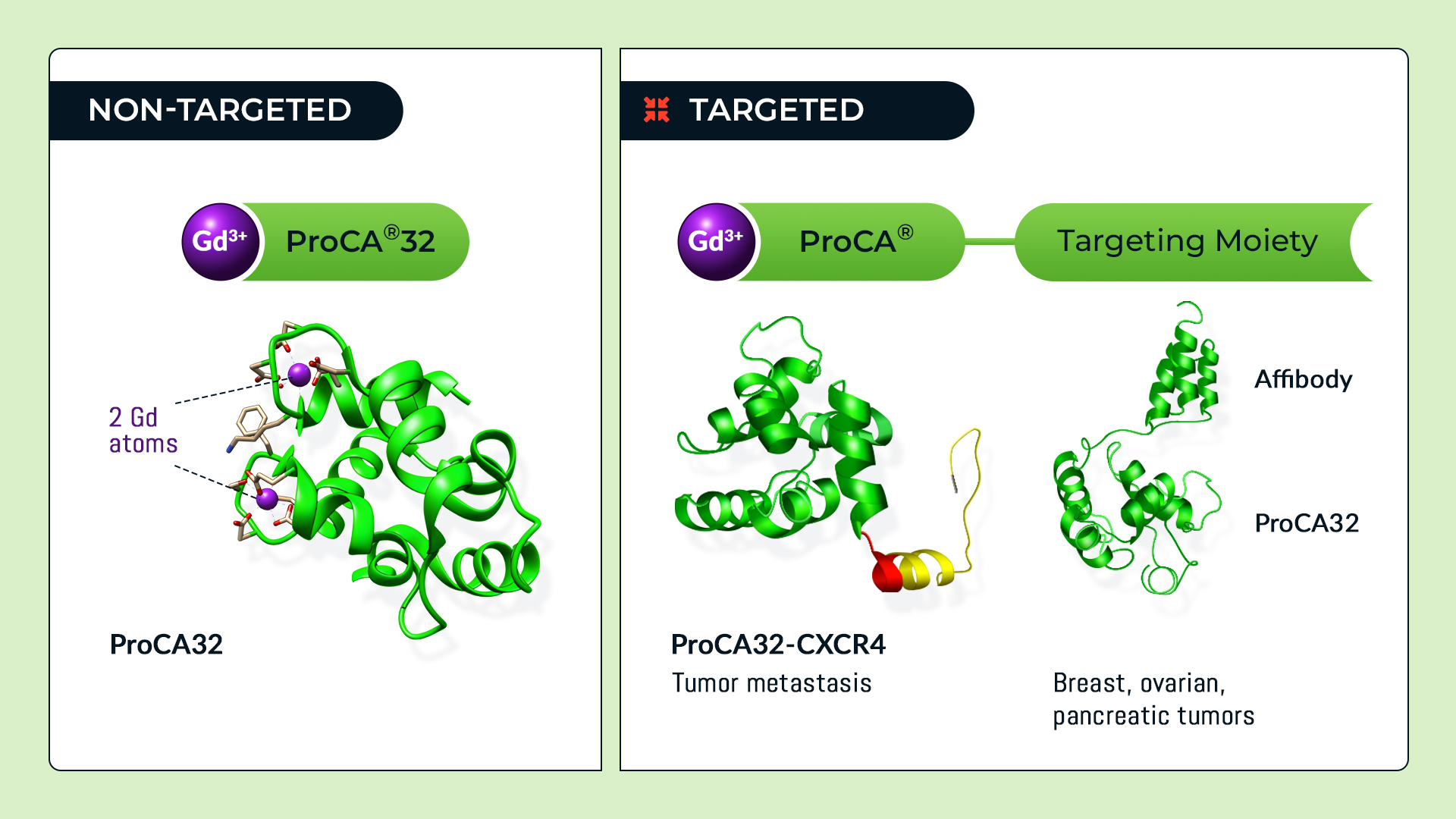
Protein Contrast Agents ( ProCA®)
InLighta has developed a novel class of protein contrast agents ( ProCA®) with significantly improved in vivo contrast capabilities. This platform technology has led to development of both blood pool (non-targeted) contrast agents for soft tissues and biomarker targeted molecular imaging contrast agents that address major unmet medical needs in non-invasive precision diagnostics.
A single injection of the blood pool non-targeted ProCA® contrast agents enables the acquisition of both bright and dark contrast imaging, which enables the early detection of small early stage tumors such as primary liver and micrometastatic lesions from breast, ovarian and uveal melanoma cancers as small as 0.1-2 mm. We have created a set of molecular imaging MRI contrast agents (tProCA) that specifically target major biomarkers including EGFR, HER2/Neu, GRPR, chemokine receptor (CXCR4), PSMA, integrin, and collagen. These targeted MRI contrast agents can detect infiltrative small lesions and distinguish benign and subtypes of cancers and their heterogeneity. We have successfully applied these targeted MRI contrast agents to non-invasively access the expression levels and patterns of biomarkers for cancer staging and to monitor disease progression or regression upon treatment in cluster 2-3 D tumor cell clusters, infiltrative tumor patterns, and xenograft murine models for prostate cancer, lung cancer, breast cancer, ovarian cancer, pancreatic cancer, liver cancer and metastasis. Collagen targeted ProCA® are able to detect early stage liver fibrosis that can be reversed with adequate treatment.
Due to the unprecedented metal selectivity of our contrast agent for gadolinium over physiological metals, we can acquire high resolution images and multi-color molecular MR imaging with low dosage, thereby significantly reducing potential issues with cytotoxicity or nephrogenic systemic fibrosis. Thus our molecular imaging contrast agents offer a safe alternative to biopsy or radioisotopes for pre-clinical and clinical applications.